STRAIN AND FERMENTATION DEVELOPMENT SERVICES
Home » Process Development » Strain and fermentation development services
YIELDS ARE NUMBER ONE
We offer you our strong expertise in developing strains based on E.coli host systems to produce peptides, proteins (soluble and insoluble), plasmid DNA products, and vaccines.
Richter-Helm BioLogics provides strain development modules for protein-based products with your choice of amino acid sequence or expression construct, or even using your candidate seed strain. Chemical and heat induction systems are available. We are also able to screen strains to determine which is most appropriate for producing your plasmid-DNA product.

Our strain development activities can be flexibly configured to suit your needs. The possibilities include:
- Construct design and cloning
- Strain screening and expression parameter testing
- Feasibility fermentation runs
- RCB production
Strain development is very closely coordinated with fermentation development, with both being handled by the experienced process development team of Richter-Helm BioLogics.
USP development at Richter-Helm BioLogics covers a wide range of leading-edge batch and fed-batch fermentation strategies based on microbial hosts such as E.coli and yeasts.
We can flexibly vary USP development components depending on your needs. The portfolio includes feasibility studies, complete USP development, and fermentation adaptation and/or optimization studies. The available general fermentation development services include:
- A process design phase performed at a small scale (e.g. of 0.3L or 1.0L) in batch or fed-batch mode
- An intermediate process scale-up to 10L
- A process engineering/adaptation phase (which can include a robustness study)
- A process reproducibility study (e.g. for scale-up purposes and/or preclinical animal studies etc.)
“Since launching our joint development project, we have been able to rely fully and consistently on Richter-Helm BioLogics.”
(customer from Pharma industry)
L
FROM PARAMETER SCREENING TO ROBUSTNESS TESTING
In addition, cell harvesting techniques such as centrifugation or crossflow filtration can be selected. Rapid at-line and offline PD analyses are available throughout the development phase, e.g.:
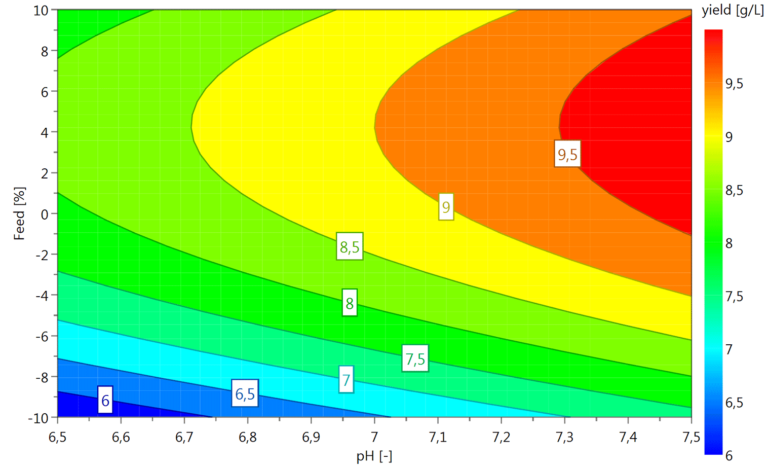
Various process parameters can be investigated to establish the most appropriate and productive fermentation approach, e.g.
- Growth rate, growth temperature, pH, DO level
- Induction mode, induction point/OD600, expression time
- Feeding profiles, media composition, etc.
- OD600, cell wet weight, cell dry weight
- Expression kinetics, EoF yield
- Substrate/byproducts
Design of experiment (DoE) tools applying full or fractional factorial design modules are routinely used to assist in developing and optimizing processes or, if applicable, to supplement parameter screenings etc.
This might interest you

USP and DSP teams work hand-in-hand to speed up process development and make every project a success.

Process characterization is key for fully understanding products and processes, establishing state-of-the-art manufacturing processes, and addressing associated process control strategies as well as any potentially critical parameters.

Richter-Helm BioLogics operates two multipurpose facilities for manufacturing biological drug substances with microbial expression systems.